There is a famous anecdote about the time Apple engineers presented Steve Jobs with an early prototype of the iPhone; Jobs complained it was too big. The engineers protested they had packed all the components as tightly as possible and could not possibly make it any smaller, at which point Jobs supposedly dropped the phone into a fish tank, pointed to the bubbles rising from it, and said, "There’s still extra space in there."
The imperative to squeeze more power into an ever-smaller form factor did not start with the iPhone, of course, but the mobile revolution has only given it more force. Tablets and "phablets" (mobile devices combining some features of tablets and smartphone handsets) offer engineers larger screens to work on than a smartphone does, but the trend toward wearable devices demands new ways to do more with less. New work on using lasers to pattern catalysts onto thin polymer sheets promises dramatically new capabilities from tiny power supplies.
Microbatteries
One such project is being carried out in the Department of Mechanical and Aerospace Engineering at the University of Missouri, where assistant professor Jian Lin, working with scientists from the U.S. Army Research Laboratory, has been using direct laser writing (DLW) to create patterns on polyimide surfaces that, given the right materials, could work as catalysts.
"Direct laser writing is a technique to control the head of a laser to make patterns," says Lin. "The technique is widely used in the packaging industry—to make designs on a bottle, for instance. We just use the commercially available equipment to do our job."
The laser generates heat and pressure where it strikes the polymer substrate. If the substrate is prepared with specific chemicals on its surface called precursors, the heat and pressure cause a localized reaction that can produce a new material. "If you choose the right precursors," Lin says, "the laser can make the materials you are interested in."
Lin and his group set out to see if the DLW process could synthesize catalysts onto a surface in desired patterns. That could point the way to producing fuel cells or microbatteries in whatever shape the computer program controlling the laser head dictated.
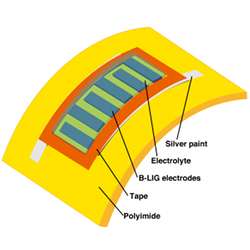
The first catalytic material Lin’s team worked with is molybdenum disulfide, MoS2. At the point where it contacts the prepared precursor, the laser produces a reaction that generates MoS2 nanoparticles anchored to carbon, resulting in a surface that is electrically conductive and which can serve as the catalyst required for micro fuel cells.
By using direct laser writing on a flexible film to create catalysts in arbitrary patterns, Lin’s team hopes to create fuel cells or microbatteries that are very thin and can take any shape needed.
Says Majid Beidaghi, assistant professor in the Department of Mechanical Engineering at Auburn University, "This work on the synthesis of catalysts indicates the power and versatility of DLW for fabrication of various types of microdevices for energy storage and conversion."
Supercapacitors
The research grew out of Lin’s postdoctoral work in the chemistry department at Rice University in Houston, TX. As part of a team led by James M. Tour, Lin helped develop a method of creating graphene using the direct laser writing technique.
"Generally, graphene is made at 1,0000C in an inert atmosphere," explains Tour. Lin’s technique uses DLW to apply heat and pressure in precise areas on a polyimide sheet. The laser carbonizes the polymer, converting its carbon to graphene and releasing nitrogen and oxygen as gases. The gas layer protects the substrate while it cools; otherwise, the hot carbon would burn up immediately when exposed to oxygen. "This is the way to make graphene in the air at room temperature," says Tour. "It leads to roll-to-roll formation, and once you can do roll-to-roll electronics, it makes things very inexpensive."
Tour’s group is exploring methods of using laser-induced graphene to make supercapacitors, which combine the high-power-burst ability of a capacitor with the long energy delivery of a battery; "that’s why everybody wants them, so you can do both," says Tour. The group has been able to produce a supercapacitor that can generate a power burst up to 25 times stronger than a lithium-ion battery, while offering the same long-term energy storage.
Tour’s team has also experimented with preparing the polyimide with chemical compounds to produce other results. "We’ve put metal salts in with the polyimide," he says, "and we’ve been able to use that material to do an oxygen-reduction reaction for a fuel cell."
Based on Lin’s and Tour’s research, future generations of wearable devices might draw their power from precisely cut pieces of plastic film, opening new possibilities in shapes and sizes.
Jake Widman is a San Francisco, CA-based freelance writer focusing on connected devices and other Smart Home and Smart City technologies.



Join the Discussion (0)
Become a Member or Sign In to Post a Comment